
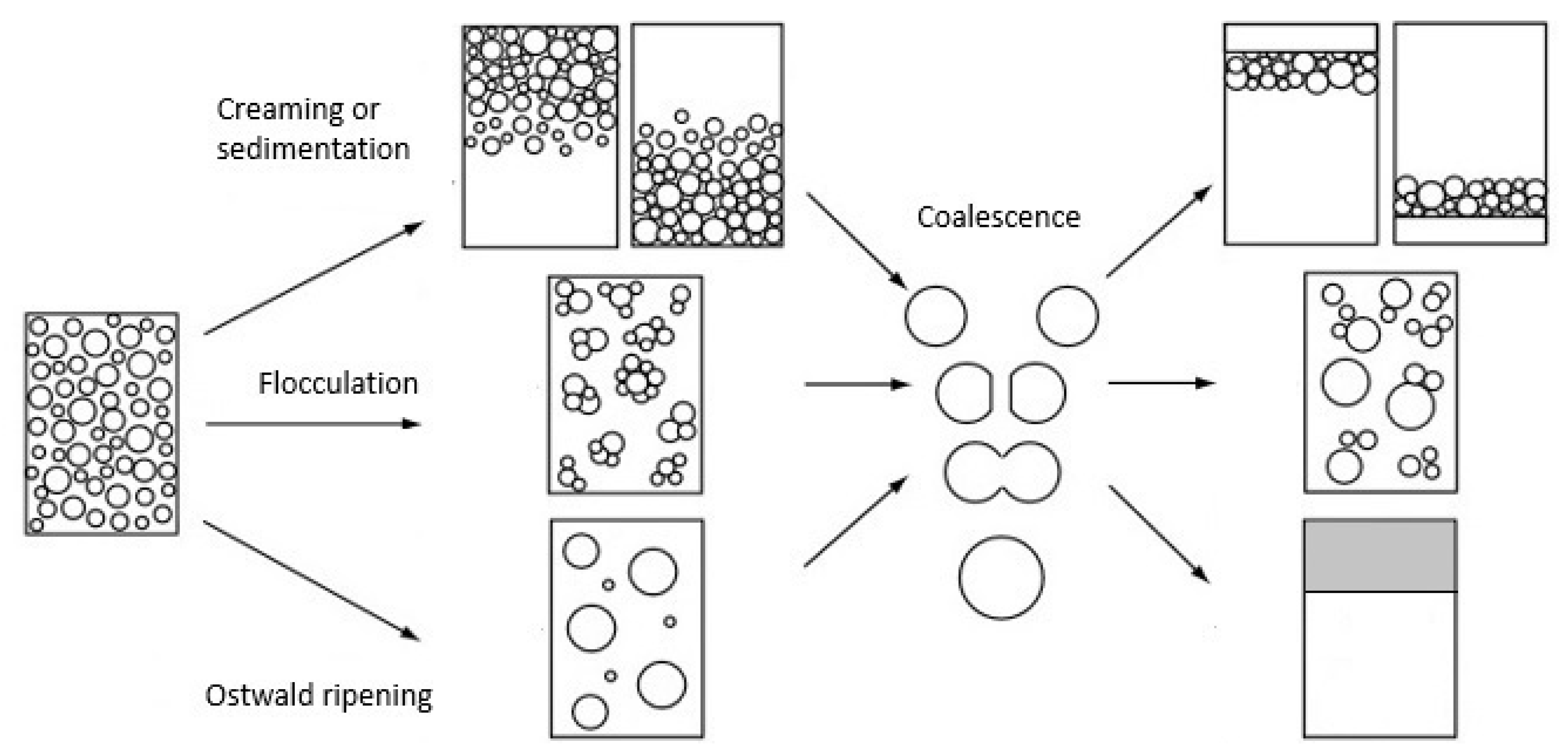
Water molecules pack closer together, so they sink to the bottom, leaving oil sitting on top of the water. That causes oil and water to form two separate layers. So what happens when you try to mix oil and water? The water molecules attract each other, and the oil molecules stick together. What happens when you mix water with oil? The oil phase is less dense than the water phase and so the oil floats on top of the water. Oil and water do not mix, instead forming two distinct layers called phases. Oil and water is a mixture, not a solution. Since oil is not soluble in water, it will never truly dissolve. Water has a low solubility when it comes to oil. These include solubilizing oils/fragrances into water, acting as primary emulsifiers, and as wetting/dispersing agents. Oil in water (o/w) emulsifiers are among the most common ingredients used in skin care products and serve a multitude of functions in formulations. An emulsion is a mixture of two or more substances that are usually immiscible. The key difference between oil in water and water in oil emulsion is that oil in water emulsions have oil droplets suspended in water whereas water in oil emulsions have water droplets suspended in oil. What is the difference between water in oil and oil in water emulsion? If you’ve never tried it before, take out a jar and combine a little bit of water and oil into the jar. When you shake or whisk the two together, they seem to be able to combine. While the word may sound technical and science-y, the concept is quite simple. The difference between oil in water and water in oil emulsion is that oil in water emulsions have oil droplets suspended in water whereas water in oil emulsions have water droplets suspended in oil. What is the difference between water in oil and oil in water? Oil and water on the other hand, form a heterogeneous or immiscible mixture. The mixture of salt and water is generally homogeneous. Solution is a homogeneous mixture of two or more components. Which type of solution is mixture of oil and water? What are some examples of oil in water emulsions?Įxamples of oil-in-water emulsions include: mayonnaise and Hollandaise sauce (featuring egg yolk lecithin as the emulsifier), homogenized whole milk, and our vinaigrette dressing (described above). (ii) Water-in-oil emulsion (W/O) : The emulsion in which water forms the dispersed phase, and the oil acts as the dispersion medium is called a water-in-oil emulsion. In milk liquid fat globules are dispersed in water. 
Milk is an example of the oil-in-water type of emulsion. What are oil in water and water in oil type of emulsion give one example of each type?
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
